|
So A is the mass number, which is equal to the number of protons, that's the atomic number which we symbolized by Z, plus the number of neutrons. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. 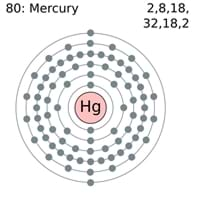
Though used for the manufacture of industrial chemicals, electrical and electronic. The mass number is the combined number of protons and neutrons in a nucleus, so it's protons and neutrons, and it's symbolized by A. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mercury, with an atomic number 80, is a heavy, d-block metal, and is the only metal liquid at standard conditions for temperature and pressure. Mårtensson, "Core-Level Binding Energies in Metals," J. Overview Mercurythe smallest planet in our solar system and nearest to the Sunis only slightly larger than Earth’s Moon. Mercury is a chemical element with the symbol Hg, named for the Latin Hydrargyrum, meaning watery or liquid silver. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. Element Mercury (Hg), Group 12, Atomic Number 80, d-block, Mass 200.592. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). Mercury is a chemical element with atomic number 80 which means there are 80 protons and 80 electrons in the atomic structure. The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. mercury atomic number 30 48 80 atomic weight 65.409 112.411 200.59 melting point (☌) 419.53 321.07 38.83. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. 
All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for mercury. These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
